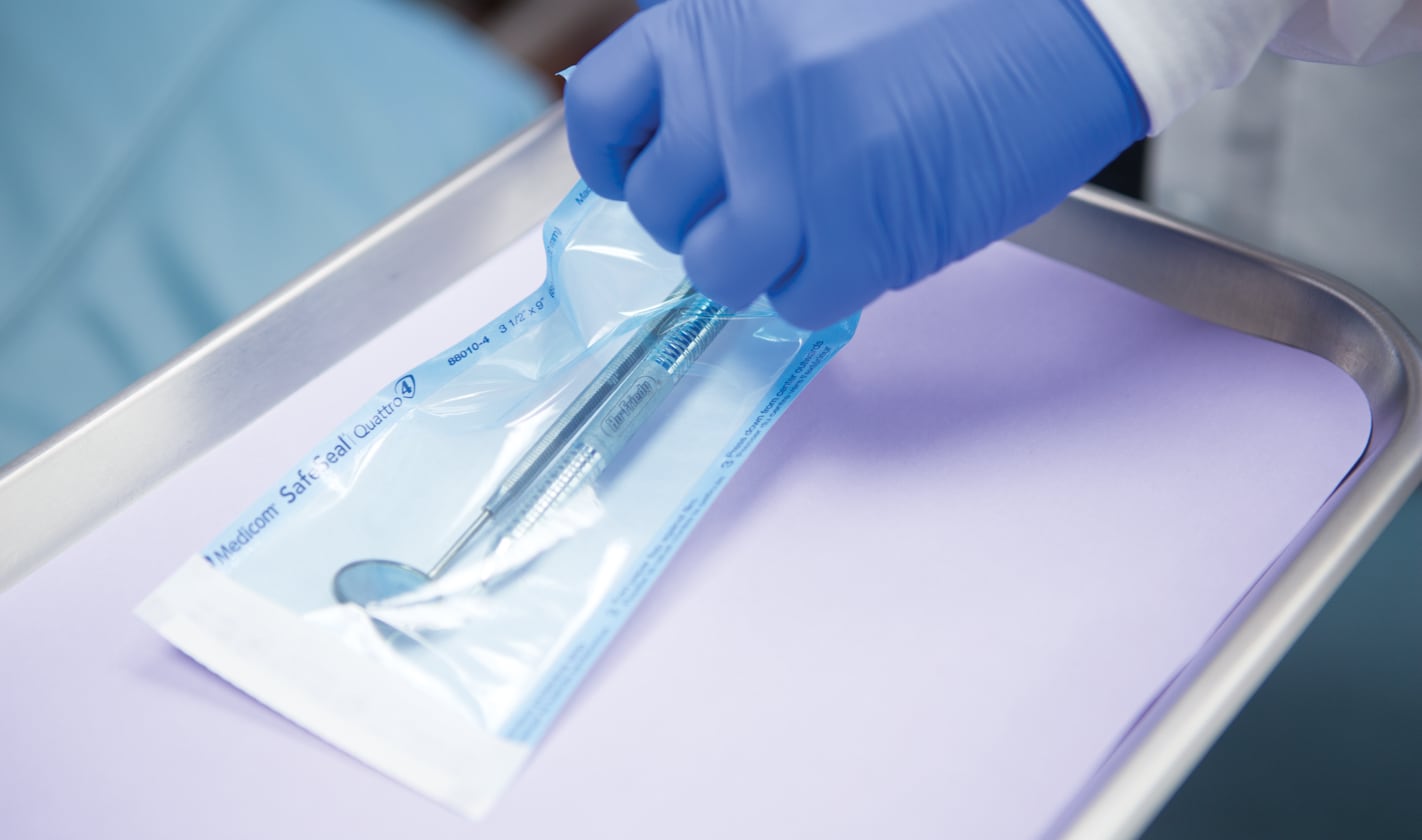
Sterilization Pouches
Sterilization pouches are single-use, peel-open pouches designed to hold items during sterilization. Not all pouch materials are suitable for use in all types of sterilization processes, so different pouches are required depending on the sterilization processes available. Plastic/paper pouches are used for steam sterilization. The plastic side of the pouch allows clear visualization of the contents and the chemical indicator. The paper side allows air removal and the sterilant to enter the pouch.
Recommended use
Sterilization pouches are recommended for smaller, lightweight, low profile items. They are available in various sizes and configurations to enclose loose groups of instruments and offer various types of built-in chemical indicators. If an instrument appears to be too heavy or large for a pouch, another packaging method should be used.
Sterilization pouches have four primary objectives:
- They must allow penetration of the chosen sterilant and be compatible with any other requirements of the sterilization process.
- They must be able to maintain the sterility of package contents until opened.
- They must create a package that can be opened without contaminating the contents by the user.
- They must permit the contents to be sterile at the point of use.
Pouches should also be “tamper evident” so that a pouch opened in error cannot be resealed without complete reprocessing.
Choosing a sterilization pouch
When choosing a sterilization pouch, look for the following features:
- The use of medical-grade Kraft paper.
- The use of a transparent polypropylene/polyester laminate film that is at least 2 mm thick.
- Adequate sizing to accommodate instruments to be sterilized without crowding.
It is important to choose sterilization pouches made from medical-grade paper rather than from recycled paper. Medical-grade Kraft paper used in sterilization pouches has a basic weight of 40-60 pounds. It contains only approved additives, has controlled porosity and has no pinholes larger than 0.5 mm. These factors are critical to ensuring that steam enters and exits the pouch only through the paper side and that the pores close during the drying process to maintain the sterility of the pouch contents.
Additional considerations may include:
- Ease of use
- Ease of storage
- Cost effectiveness
- Puncture and tear resistance
- Toxic ingredients or non-fast dyes?
- Non linting material
Selecting pouch size
It is important to select the appropriate pouch size. A pouch should be large enough to allow the contents to move from side-to-side without compromising the seams or risking punctures. A pouch that is too small may also cause overcrowding that prevents adequate steam penetration and air removal, which could affect drying of instruments. Conversely, a pouch that is too large may allow excessive movement of the instruments that results in punctures and tearing. For that reason, it is a good idea to have a variety of pouch sizes on hand.
Loading a sterilization pouch
To load a sterilization pouch, the pouch should be laid flat on a countertop and the instruments should be arranged in a single layer with at least ¼ of an inch of clearance on each side for package contraction and proper circulation.
To allow easy removal and ensure aseptic presentation of the instruments when opening the pouch, position instruments in the pouch as follows:
- Place instruments with handles toward the pre-sealed end of the pouch.
- Single-ended instruments should be positioned in a pouch with the handle/grasp or nonworking end toward the thumb notch and away from the adhesive-sealed end.
- Hinged instruments should be placed in an open position.
- Curved instruments should be placed with the curved end toward the plastic side of the pouch to avoid punctures though the paper side.
- On instruments with sharp tips, use clear-vented tip protectors to prevent punctures through the pouch.
Sealing a sterilization pouch
Before sealing, excess air should be removed from the pouch. Sterilization pouches are either self-sealing or manually heat-sealed. When using a self-sealing pouch, it is important to carefully follow the fold lines provided by the manufacturer to prevent overlapping and small openings between the package and the sealant. Also, the seal’s fold should be free of wrinkles or creases.
When manually sealing a pouch, ensure that there is ample space at the end of pouch for users to use their thumb to open the package and present its contents aseptically to the sterile field. This will prevent the contamination of the pouch contents during delivery.
When using a heat-sealed pouch, it is important to follow the manufacturer’s instructions, including proper temperature ranges.
Loading pouches into a sterilizer
When placing loaded and sealed pouches in the sterilizer, it is important to ensure that they do not overlap. Ideally, pouches should be placed on their edges on a rack to allow 360-degree circulation and penetration of steam and accelerate drying. To prevent tearing, pouches should not be touched until they are completely dry.
Pouch labeling
At a minimum, sterile pouches should be labeled with the following information: sterilizer used, cycle/load number, date of sterilization and expiration date if applicable.
To write on pouches, be sure to use only non-toxic permanent markers approved for the sterilization process (ASTM D4236) and to write only on the plastic laminate of the pouch, as writing on the paper could compromise the integrity of the pouch.
Chemical indicators
International Organization for Standardization (ISO) standards for sterilization packaging performance stipulate that the only way to be sure an item has been effectively steam-sterilized is to expose it to all three sterilization variables: time, temperature and the presence of steam. However, many sterilization pouches only have an external Type 1 process indicator that only validates exposure to heat (temperature). More advanced pouches, like Medicom SafeSeal® Quattro self-sealing sterilization pouches, have both internal and external multiparameter indicators, which confirm that all three criteria for sterilization have been achieved and that the contents have been exposed to the critical variables.
Storing sterilized pouches
Proper storage and handling of processed pouches is necessary to maintain the sterility of their contents. Storage room temperature should be 75°F (24°C) or less with humidity less than 70%.
The CDC recommends that sterile instruments and supplies be stored in covered or closed cabinets and not under sinks or in other places where they might become wet. Pouches should be inspected before opening and use to ensure that the packaging has not been compromised (i.e., wet, torn, or punctured) during storage.
Shelf life of processed pouches
The shelf life of a processed sterilization pouch is event-related, not time-related, and depends on proper handling and storage. It also depends on the quality of the pouch material, the conditions under which the pouch is stored and the amount of handling. A sterile product will remain sterile as long as the packaging does not become wet, torn, soiled or damaged.
It is important to note that all sterilization pouches list a manufacturer’s materials expiration date and may also list a shelf-life date. For example, a pouch may have a three-year from date of manufacture expiration date and a two-year expiration from date of sterilization. This information is critical to stock rotation and a using sterilization pouch past its expiration date is considered off-label use by the FDA.
Why choose Medicom SafeSeal® Quattro self-sealing sterilization pouches
Medicom SafeSeal® Quattro self-sealing sterilization pouches are crafted in our own state-of-the-art manufacturing facilities. They are mechanically sealed using TruePress™ technology, which fuses the proprietary, blue-tinted film to high-grade medical paper to form a perfect bond that ensures a leak-proof seal that protects the integrity of your instruments.
Medicom SafeSeal Quattro sterilization pouches confirm sterilization parameters − time, temperature and steam/EO − with internal and external multi-parameter process indicators printed directly on the pouch.


For larger items, Medicom® peelable sterilization reels are available in various sizes for customized sterilization of a wide variety of items. Medical grade paper provides superior strength. Blue-tinted transparent material allows easy viewing of pouch contents to identify possible breaches.
For more information about Medicom sterilization products, visit our website.

To learn more about the sterilization process, sign up for our FREE CE course, “Instrument Sterilization: Because Patient Safety Matters”.



 English (US)
English (US)